
A butadiene molecule contains four carbon atoms.

(a) How are the carbon atoms hybridized? sp? sp²? sp³?
(b) Where are the σ-bonds and π-bonds located in this molecule?
(c) How coud the energies of the molecular orbitals be calculated?
(a) sp².
(b) The two C=C double bonds consist of a single σ and a π bond. All other bonds are σ bonds.
(c) A trial molecular orbital wavefunction is constructed as a linear combination of the partially filled valence orbitals. In this case it would the four carbon pz orbitals.
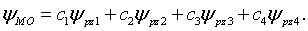
The energy of this the molecular orbital is,
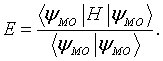
The energy is minimized with respect to the coefficients, ci. The four equations that result from this minimization are,
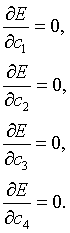
These equations can be expressed in terms of the matrix elements of the Hamiltonian. In the Hückel model, the resulting matrix equation is,
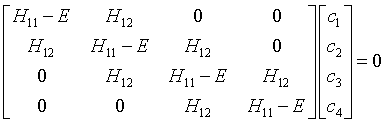
The eigen values of this matrix are the molecular orbital energies.