PHY.K02UF Molecular and Solid State Physics
|
| ||||
PHY.K02UF Molecular and Solid State Physics | ||||
Calculating a bond potential was discussed in the molecules section of the online notes. This is a computationally intensive calculation. Often a bond potential is fit to a simple function. For van der Waals bonds, a Lennard-Jones potential is often used. Van der Waals bonds are weak bonds that appear in molecular crystals (like sugar crystals) and the solids of ideal gas elements that form at low temperature. For covalent bonds, the Morse potential is often used.
5.x The bond between atoms bound by a van der Waals bond can be described by a Lennard-Jones potential,
\begin{equation} U(r)= U_0\left(\left(\frac{r_0}{r}\right)^{12}-2\left(\frac{r_0}{r}\right)^{6}\right). \end{equation}If $\epsilon = 1.02\times 10^{-21}$ J, and $\sigma= 0.312$ nm, what is the bond length $r_0$ that minimizes the energy?
For small deviations around $r_0$, the force between the two atoms can be described by Hooke's law $F=-k(r - r_0)$. Here $k$ is the effective spring constant. What is the effective spring constant for this bond?
5.1 The bond between two atoms can be described by a Morse potential where $U_0 = 3 \,\text{eV}$, $r_0 = 0.16 \,\text{nm}$, and $a = 0.08\,\text{nm}$.
\begin{equation} U(r)= U_0\left(e^{-2(r-r_0)/a}-2e^{-(r-r_0)/a}\right). \end{equation}For small deviations around $r_0$, the force between the two atoms can be described by Hooke's law $F=-k(r - r_0)$. Here $k$ is the effective spring constant. The relationship between the potential and the force is $F = -\frac{dU}{dr}$. What is the effective spring constant for this bond?
The atoms form a crystal 1 cm high, 1 cm wide, and 6 cm long. The crystal has a simple cubic structure and the bond between each pair of atoms can be described by the Morse potential given above. The crystal is stretched along the long axis. What is the spring constant of the crystal?
Ionic crystals are held together predominantly by long range Coulombic forces. It is necessary to sum the Coulomb energies over the nearest neighbors atoms and also the more distant neighbors to calculate the energy correctly. Ionic bonds are typically a few eV.
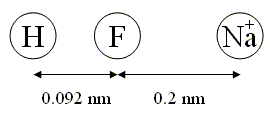
5.2 The molecule HF (hydrofluoric acid) shows a bond length of 0.092 nm. The molecule is characterised by a very strong partial charge transfer of the electron from hydrogen to fluorine of 0.41 $e$. A sodium ion (Na+) is 0.2 nm away from the HF molecule. Calculate how much energy it would take to move the Na+ far to the right of the molecule. Assume that the atoms remain in a line.
5.x Explain what the Madelung constant is.
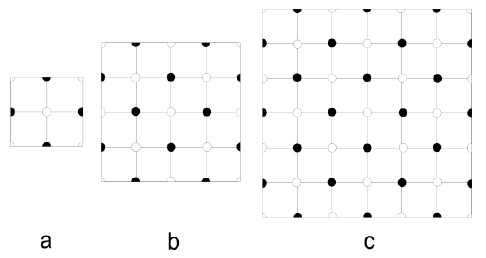
5.3 Determine the Madelung constants of the different sections of the two dimensional ionic crystal in the figure below. Compare the results with the Madelung constants of the infinite crystal $(\alpha = 1.6155)$. Why are the sections selected with the atoms at the border of the sections truncated?
5.4 The cohesive energy of a crystal is the energy needed to separate all of the atoms. The cohesive energy of NaCl can be described by the function,
\begin{equation} U_{tot}= N\left(z\lambda e^{-R/\rho}-\frac{\alpha e^2}{4\pi\epsilon_0 R}\right). \end{equation}Here $N$ is the number of primitive unit cells in the crystal, $R$ is the interatomic spacing, $z$ is the number of nearest neighbors, $\lambda,\rho$ and $\alpha$ are constants. For NaCl, $\rho = 0.321$ Å, $z\lambda = 1.05\times 10^{-15}$ J, and $\alpha = 1.747$.
(a) Determine the lattice constant of the conventional unit cell from this expression.
(b) Calculate the effective spring constant of a single bond.
(c) Calculate the bulk modulus $B = -V(dp/dV)$ and the compressibility $\kappa = 1/B$ of an ideal sodium chloride crystal at $T = 0$ K. Here $V$ is the volume and $p$ is the pressure. At $T = 0$ K, the entropy is zero so the total cohesive energy is $dU_{tot} = -p dV$. This means that the bulk modulus at low temperature is,
\begin{equation} B= V\frac{d^2U_{tot}}{dV^2}. \end{equation}5.x The CsCl structure has the largest Madelung constant and therefore ionic crystals forming this structure have the lowest energy. Explain why some ionic crystals form the NaCl or Zincblend crystal structures instead of the low energy CsCl structure.
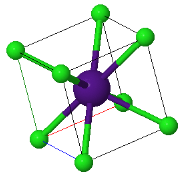 | 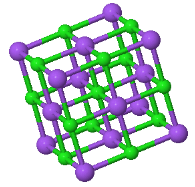 | 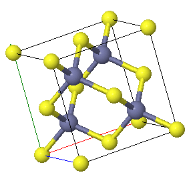 |
CsCl | NaCl | Zincblend |